Greater Awareness Needed of Delayed Skin Reactions to Moderna COVID-19 Vaccine
COVID19 - CORONAVIRUS, 8 Mar 2021
Eurasia Review - TRANSCEND Media Service
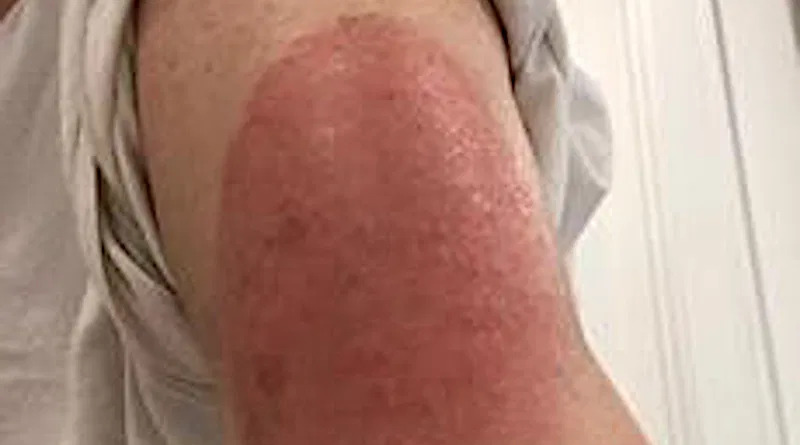
Published in The New England Journal of Medicine: Researchers from Massachusetts General Hospital note that Phase 3 clinical data from the Moderna vaccine trial did show delayed skin hypersensitivity in a small number of the more than 30,000 trial participants. However, the authors say the large, red, sometimes raised, itchy or painful skin reactions were never fully characterized or explained, and they warn clinicians may not be prepared to recognize them and guide patients on treatment options and completion of the second dose of the vaccine.
CREDIT: Massachusetts General Hospital
4 Mar 2021 – As the speed and scale of vaccinations against the SARS-CoV-2 virus ramps up globally, researchers at Massachusetts General Hospital (MGH) are calling for greater awareness and communication around a delayed injection-site reaction that can occur in some patients who have received the Moderna mRNA-1273 vaccine.
In a letter to the editor published online in The New England Journal of Medicine (NEJM), the authors note Phase 3 clinical data from the Moderna vaccine trial did show delayed skin hypersensitivity in a small number of the more than 30,000 trial participants. However, the authors say the large, red, sometimes raised, itchy or painful skin reactions were never fully characterized or explained, and they warn clinicians may not be prepared to recognize them and guide patients on treatment options and completion of the second dose of the vaccine.
“Whether you’ve experienced a rash at the injection site right away or this delayed skin reaction, neither condition should prevent you from getting the second dose of the vaccine,” says Kimberly Blumenthal, MD, MSc, lead author of the letter and co-director of the Clinical Epidemiology Program in the division of Rheumatology, Allergy and Immunology at MGH. “Our immediate goal is to make physicians and other care providers aware of this possible delayed reaction, so they are not alarmed, but instead well-informed and equipped to advise their patients accordingly.”
In the letter, Blumenthal and her co-authors also note their own clinical observations of the delayed, large, local reactions to the Moderna vaccine, and report on a series of 12 patients with the reactions. In that group, symptom onset ranged from four days after the first dose up to 11 days post-vaccination, with a median onset of symptoms on day eight. Photographs show the varied size and severity of the reactions. Most patients were treated with ice and antihistamines, although some required corticosteroids and one was erroneously treated with antibiotics.
“Delayed cutaneous hypersensitivity could be confused – by clinicians and patients alike – with a skin infection,” says letter co-author Erica Shenoy, MD, PhD, associate chief of the MGH Infection Control Unit. “These types of reactions, however, are not infectious and thus should not be treated with antibiotics.”
On average, symptoms cleared up after nearly a week for the group of 12 reported in the letter. Half of the patients went on to experience a reaction after the second dose – at or around 48 hours post-vaccination. No patient experienced a dose two reaction that was more severe than their dose one reaction.
The authors also say samples taken from skin biopsies confirmed their suspicion of a delayed allergic immune response that is commonly seen in drug reactions.
“For most people who are experiencing this, we believe it’s tied to the body’s immune system going to work,” says Esther Freeman, MD, PhD, director of Global Health Dermatology at MGH and co-author of the NEJM letter. “Overall, this data is reassuring and should not discourage people from getting the vaccine.”
Go to Original – eurasiareview.com
Tags: COVID-19, Coronavirus, Vaccines
DISCLAIMER: The statements, views and opinions expressed in pieces republished here are solely those of the authors and do not necessarily represent those of TMS. In accordance with title 17 U.S.C. section 107, this material is distributed without profit to those who have expressed a prior interest in receiving the included information for research and educational purposes. TMS has no affiliation whatsoever with the originator of this article nor is TMS endorsed or sponsored by the originator. “GO TO ORIGINAL” links are provided as a convenience to our readers and allow for verification of authenticity. However, as originating pages are often updated by their originating host sites, the versions posted may not match the versions our readers view when clicking the “GO TO ORIGINAL” links. This site contains copyrighted material the use of which has not always been specifically authorized by the copyright owner. We are making such material available in our efforts to advance understanding of environmental, political, human rights, economic, democracy, scientific, and social justice issues, etc. We believe this constitutes a ‘fair use’ of any such copyrighted material as provided for in section 107 of the US Copyright Law. In accordance with Title 17 U.S.C. Section 107, the material on this site is distributed without profit to those who have expressed a prior interest in receiving the included information for research and educational purposes. For more information go to: http://www.law.cornell.edu/uscode/17/107.shtml. If you wish to use copyrighted material from this site for purposes of your own that go beyond ‘fair use’, you must obtain permission from the copyright owner.
Read more
Click here to go to the current weekly digest or pick another article:
COVID19 - CORONAVIRUS: